Carbon electron configuration standard form10/19/2023  Both of the configurations have the correct numbers of electrons in each orbital, it is just a matter of how the electronic configuration notation is written ( here is an explanation why). Note that when writing the electron configuration for an atom like Cu, the 3d is usually written before the 4s. Therefore the expected electron configuration for Copper will be 1s 22s 22p 63s 23p 64s 23d 9. After the 4s is full we put the remaining six electrons in the 3d orbital and end with 3d9. We now shift to the 4s orbital where we place the remaining two electrons. Since the 3s if now full we'll move to the 3p where we'll place the next six electrons. We'll put six in the 2p orbital and then put the next two electrons in the 3s. The p orbital can hold up to six electrons. The next six electrons will go in the 2p orbital. Since 1s can only hold two electrons the next 2 electrons for Copper go in the 2s orbital. In writing the electron configuration for Copper the first two electrons will go in the 1s orbital. The element is in the 2nd column of the p block, Group IVA (Column 13).Video: Cu, Cu +, and Cu 2+ Electron Configuration Notation Germainum is in the 4th row Energy Level of the periodic table. The d orbitals Groups 3-12 (columns) can hold 10 electrons.Įach energy level must be filled before moving up an energy level.Įach orbital group must fill before moving to the next orbital group. The p orbitals Groups 13 - 18 (columns) can hold 6 electrons The s orbitals Groups 1 & 2 (columns) can hold 2 electrons The superscript tells us the number of electrons in the orbital. The Coefficient tells us the Energy Level (Row) of the periodic table The electron configuration for the first 10 elements The "f block" on the periodic table are the Lanthanide and Actinide series.Įlectron Configurations are an organized means of documenting the placement of electrons based upon the energy levels and orbitals groupings of the periodic table. The "d block" on the periodic table are groups 3-12 make up the d block and the elements' electron configurations end in d. The "p block" on the periodic table are groups 13-18 and end in p1, etc. 
The "s block" on the periodic table are groups 1 and 2 they end in s1 and s2. These rare earth metals are 2 periods behind because the f electrons are even higher in energy than the d electrons. The transition metals are behind by one period because the d electrons are high in energy.įor the rare earth elements (the Lanthanides and Actinides), they end in f. Scandium would end in 3d1, titanium in 3d2, etc. The general rule is that the element's electron configuration ends in d and whatever place they are in. And so it goes.įor the transition metals, groups 3-12, there are many exceptions. In group 4A or 14, all elements end in p2. Group 3A, or 13 all end their electron configurations in p1. Group 2 elements (2A), the alkaline earth metals, all end in s2 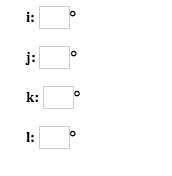
What period the element is in determines the 1st number.Įxample: H ends in 1s1 (even though H is not a metal, it resides in this group because it also has one valence electron) Group 1A (1), the alkali metals all end is s1. When looking at electron configuration, your fill order of electrons is: 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
